



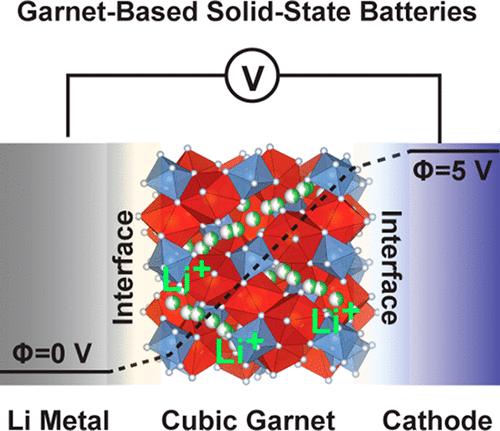
All-Solid-State Batteries (ASSBs) that use oxide-based solid electrolytes (SEs) have been considered as a promising energy-storage platform to meet an increasing demand for Li-ion batteries (LIBs) with improved energy density and superior safety. However, high interfacial resistance between particles in the composite electrode and between electrodes and the use of Li metal in the application specific batteries hinder their practical utilization. The research progress is on oxide-based SEs for the ASSBs with respect to the use of Li metal. We especially focusing on research progress on garnet-type solid electrolytes (Li7La3Zr2O12) solid ionic conductor and their doped variant towards the development of all solid state batteries because they have high ionic conductivity, good chemical stability with Li metal, and a wide electrochemical potential window.